Testing Plant Substances as Potential Medicines
Background: All organisms are infected by viruses and diseases and many of those organisms have developed ways to battle them. Finding and isolating the antimicrobial molecule that are sometimes present in plants could help us make medicine to treat diseases in humans. Isolating an active ingredient that has antimicrobial activity is a complicated and important process. The end results can include antiseptics, astringents, antibiotics, and toxins.
Purpose:
What plant materials, found locally, contain active ingredients that will inhibit the growth of bacteria?
Materials:
Balance, weight boat, lab scoops
LB broth base
Media bottles, 250 mL
Sterilizer/autoclave
Water bath, 37 degrees Celsius, shaking
Sterile LB agar
Laminar flow hood an
Filter paper disinfectant
Glasses, safety, plastic
Bunsen burner and gas lighter
Inoculating loop, Ni/Cr wire
Petri dishes, 60x15 mm , sterile
E. coli JM109 (stock plate)
Plant specimen
Mortar and pestle
Pipet, 10 mL and pump
Plastic funnels, short-stemmed
Filter paper disks, 5 mm diameter
Beakers, 100 mL
Syringe, 10 mL and filter, 0.2 mm
Reaction tubes and rack, 1.7 mL
Methanol, absolute
Pipet, 1 mL and pump
Dry block heater/heat block
Forceps, fine-tipped
Ampicillin
Glass spreader
Incubator oven, 37 degrees Celsius
Procedure:
Filter Sterilization:
The profilter was attached to a syringe and “rinsed” with H2O. Next the sterilization filter was opened, with the filter kept in its plastic covering. Approcimately 1.5 mL of H20-based filtrate was loaded using a pipet. The plunger was used to collect the sterile filtrate into the microfuge tubes. The microfuge tubes were then quickly closed until a "snap" sound was heard.
Preparing Plant Extracts:
The mortar and pestle was used to grind up 2 grams of plant tissue with 10 mL of deionized water. The deionized water was let sit for 3 minutes and the sample was filtered through an 11 cm filter paper funnel. The filtered sample extract was filter sterilized using a syringe filter. 1 mL of extract was collected into a 1.7 mL microtube. These steps were then repeated, but the water was replaced with methanol as the extracting solvent.
Final Procedure:
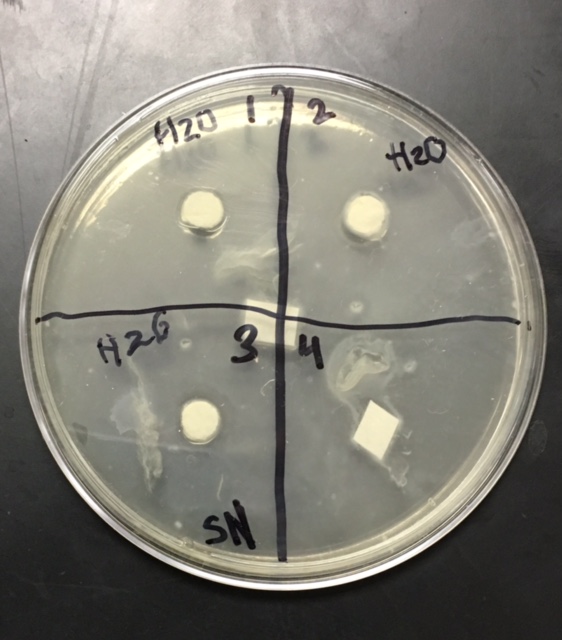
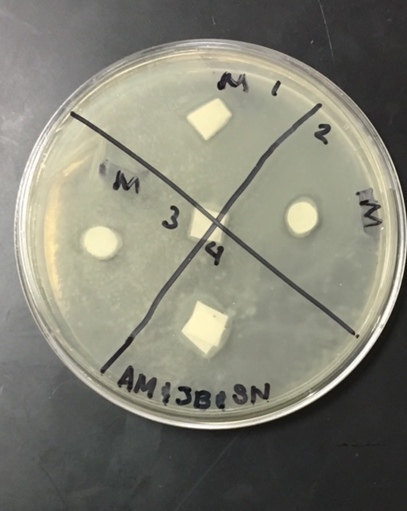
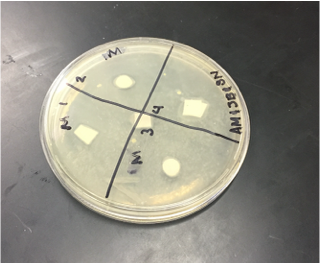
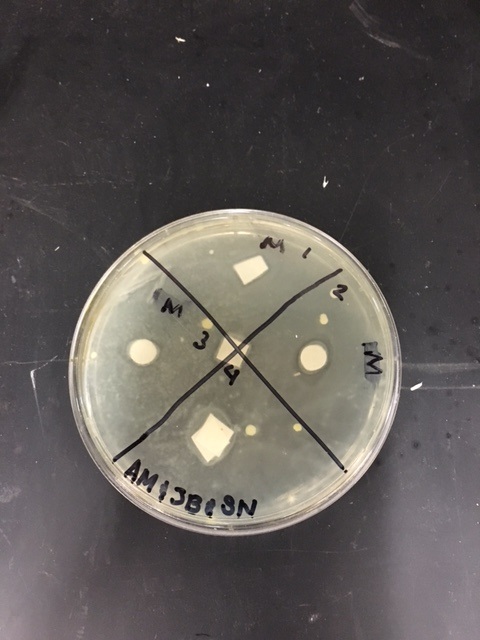
Sterile foreceps were used to add sterile disks to tubes containing 1 mL of sterile water and 1 mL of ampicillin. 10-20 mLs of warmed, pressure sterilized nutrient agar was poured into 2 petri dishes, using sterile technique. After allowing the agar to solidify, plates were turned upside down and sterilized at 4º Celsius overnight. One mL of E.coli colony was added to each plate. A flame-sterilized spreading loop was used to spread the bacteria evenly throughout the surface of the agar. Using sterilized forceps, filter disks were placed in separate quadrants onto the plate in the following sequence: 1) water 2) plant extract 3) ampicillin. Plates were left on the lab bench for 20 minutes to allow bacteria and filter disks to adhere to the agar. Plates were then incubated upside down overnight in a 37º Celsius incubator. Plates were then photographed and observed for clearings around the filter disks after 24 hours, 48 hours and 72 hours. Any bacterial or fungal contamination was noted.
Observations:
After the first 24 hours we noticed some bacterial growth that was obviously contamination in our H2O petri dish. This could only have occurred if the plate was contaminated during one of the steps of our procedure, and was most definitely a human error. In both of our dishes we could observe clearance in our ampicillin quadrant.
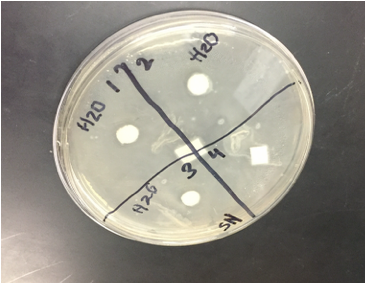
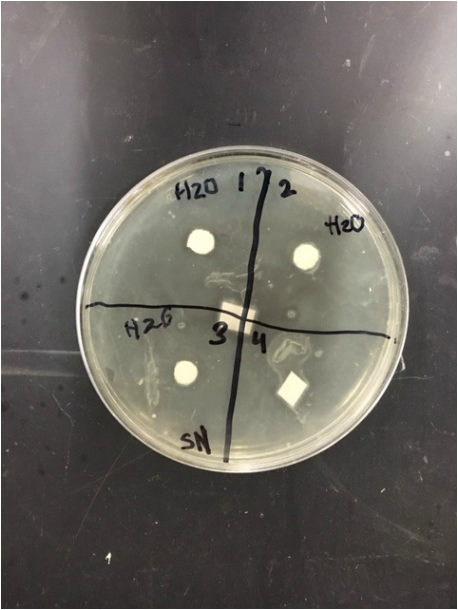
When we observed our petri dishes after 72 hours, we could see some contamination in the form of bacteria in both our H2O dish and our methanol dish. This was disappointing, but since this is science in practice and not just in theory, one can only expect to have some results that are subject to human error.
During our 72 hour observation our group could see continued growth in the bacterial contaminations and that there continued to me clearance in every quadrant 4.
Analysis/Conclusion: The positive control disk, containing the ampicilin, was the only disk to have any major clearance around it, as it was most likely the only compound in the petri dish to have any antimicrobial properties. The bacterial lawn had been contaminated, and my group found it hard to determine whether the clearance around the ampicilin was effected by the unintended bacterial growth. The negative control (the water disk) had no clearing. It was very difficult to interpret this disk, due to human error while performing the procedure. Obviously, water would not have antimicrobial properties because it had no chemical or biological compounds that could deter, harm, or disable any known bacteria and that is why it is used as the control group in this experiment. Our plants did not have strong antimicrobial properties as far was could be discerned from this trial. It would be necessary to perform this experiment many more times to get more accurate results, especially since our disks were contaminated by unintended bacterial growth.